Extended Depth of Focus IOL (EDOF IOL)
Home / Guide to Cataract Surgery /
Last Updated:
If you are getting cataract surgery or refractive lens exchange (RLE), an extended depth of focus or EDOF IOL is a premium lens option to consider. EDOF lenses offer a wide range of seamless clear vision, offering spectacle independence for most everyday tasks.
Table of Contents
What Are Extended Depth of Focus (EDOF) IOLs ?
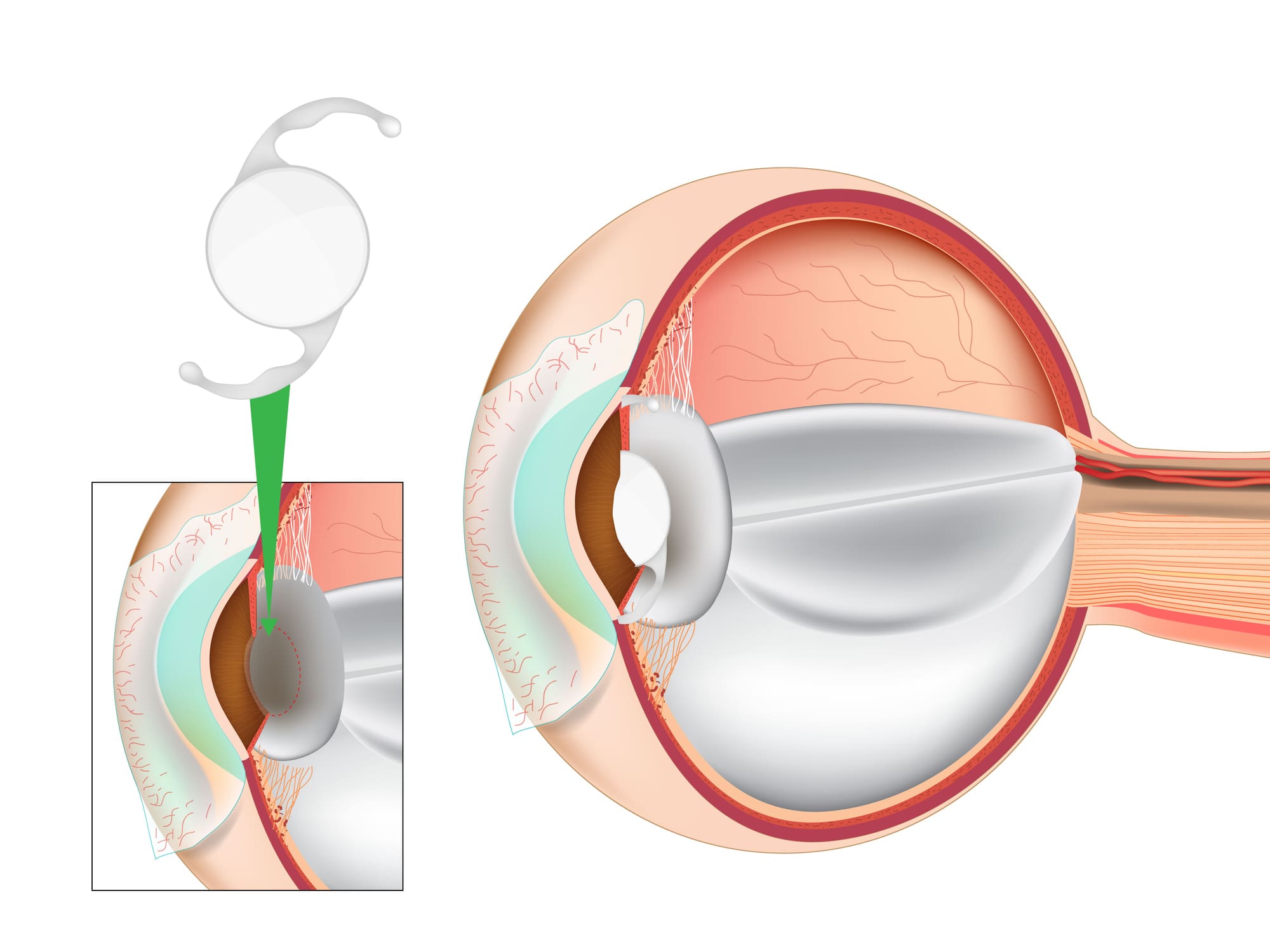
Extended Depth of Focus (EDOF) intraocular lenses are a special kind of lens used in cataract surgery or refractive lens exchange (RLE). They work by manipulating light to create a single, elongated focal point to enhance the depth-of-focus (rather than having multiple discrete focal points like multifocal IOLs). Clear vision is continuous across the range, though vision quality is stronger for intermediate and distance vision.
EDOF IOL vs Multifocal IOL
EDOF intraocular lenses are sometimes referred to as multifocal IOLs. This is because both lenses provided good vision at near, intermediate and distance ranges. However, they utilize differnt technologies to achieve this. The distinction is important when considering your options for cataract surgery or RLE.
You deserve clear vision. We can help.
With 135+ locations and over 2.5 million procedures performed, our board-certified eye surgeons deliver results you can trust.
Your journey to better vision starts here.
How Do EDOF IOLs Work?
Extended depth of focus IOLs use different mechanisms to create an extended range of vision. EDOF IOLs currently available for commercial use incorporate one or more of the following technologies:
Spherical Aberration
Some EDOF lenses manipulate light to enhance depth of focus through “spherical aberration”. This means that light rays entering the lens at different angles will focus at slightly different points, creating a broader range of clear vision. When spherical aberration occurs naturally in the eye, the resulting image tends to be blurry. However, EDOF IOLs are engineered to take advantage of these aberrations to improve overall visual performance. Spherical aberration is combined with other mechanisms that shape and control how light enters the eye and the way it behaves.
If the magnitude of the aberrations is too large, distance image quality will be compromised. For this reason, many models that use spherical aberration incorporate the pinhole effect to enhance vision outcomes.
Pinhole Effect
This technology consists of a small aperture or “pinhole” in the IOL design. This pinhole blocks light rays that are not focused, particularly those that enter at angles that would typically cause blurriness. By allowing only paraxial (central) light rays to pass through, the lens effectively reduces the impact of defocused light.
The smaller the aperture, the greater the depth of field (the range within which objects appear sharp). This allows patients to maintain clarity when viewing both near, intermediate and far objects.
This design helps eliminate the overlapping of near and far images. While this can improve clarity and visual transitions, it may reduce vision quality in low-light conditions due to decreased light transmission.
Patients with naturally large pupils might want to consider a different style of EDOF lens, as they are more likely to experience visual disturbances and a reduced depth of focus with pinhole-effect models.
Diffractive Technology
Some EDOF IOLs use the same technology as multifocal IOLs to create multiple focal points. The focal points are then elongated to enhance depth of focus and “connect” the focal points, creating a continuous field of clear vision. However, EDOFs that use this technology – like the Tecnis Symfony – are associated with increased levels of visual disturbances.
Beam-Shaping Technology
More recent EDOF lenses, like the AcrySof IQ Vivity, use wavefront-shaping (beam-shaping) technology to achieve enhanced vision results. This mechanism was designed to provide the vision results expected of EDOF lenses without the visual disturbances and decreased vision quality associated with older models.
Wavefront-shaping technology redistributes light to elongate the focus, enhancing vision throughout an expanded range while minimizing visual disturbances like halos and glare that are common with multifocal lenses. These lenses maximize the efficiency of light across the near to distance vision range.
Who is an Ideal Candidate for EDOF Lenses?

EDOF IOLs are ideal for patients who require good intermediate vision and desire a broader range of clear vision without the need for glasses. Tasks that require good intermediate vision include computer work, crafts and hobbies like knitting or sewing, board games, cooking and certain social interactions, like a conversation with a group of people gathered around a table.
Keep in mind that while near vision will also be improved, it will not be as clear as with monofocal or even some multifocal IOLs.
Who is Not a Candidate for EDOF Lenses?
Not everyone is a good candidate for EDOF IOLs. The presence of any of the following conditions could make you a poor candidate for these lenses, and should be discussed with your doctor before you settle on an EDOF IOL:
- Fuch’s dystrophy
- History of post-refractive surgeries
- Advanced dry eyes
- Anterior basement dystrophy
- Weak zonules
- Glaucoma
A discussion with your eyecare provider about IOL options is highly recommended prior to cataract surgery or RLE, even if you have none of the conditions mentioned on the list. Based on your lifestyle, medical history and preferences, your doctor might recommend a different IOL.
You deserve clear vision. We can help.
With 135+ locations and over 2.5 million procedures performed, our board-certified eye surgeons deliver results you can trust.
Your journey to better vision starts here.
Extended Depth of Focus Pros and Cons
The main advantage of an extended depth of focus IOL is that clear vision will be continuous throughout. Patients can benefit from a wide range of vision with smoother transitions between near, intermediate and distance focal points. However, they should not expect the same quality of vision as when they were younger. No IOL can match the vision power of a healthy eye with a natural lens, but vision outcomes can still be very good.
EDOF lenses provide excellent distance vision that rival traditional monofocal lens implants and great intermediate vision (e.g. computer or car dashboard).
In terms of near vision, EDOF lenses typically provide only the equivalent of a mild reading eyeglass power. Therefore, reading eyeglasses may still be needed for small print.4 Reading glasses may be also needed for patients that like to hold their books close to them.
EDOF lenses may also induce more glare and decrease contrast sensitivity than other lenses, especially in dimly lit environments such as night driving.
EDOF IOL vs Other Lenses
EDOF IOL vs Monofocal IOL
Traditional monofocal lens implants can only correct one distance: distance or near (although less commonly, the IOL can be focused for intermediate vision). EDOF IOLs, by contrast, improve visual acuity at close, intermediate and far ranges, although near vision will be less sharp.
Patients who are not willing to compromise on quality near vision might prefer a monofocal IOL, though they will require lenses to see clearly at other distances. EDOF lenses also tend to induce more glare than a traditional monofocal IOL.
EDOF IOL vs Multifocal IOL
As mentioned, EDOF lenses tend to show superior vision for distance and intermediate but may be less clear for near targets. Multifocal IOLs have gotten better at providing great quality of vision at different focal points. The most advanced model, the PanOptix trifocal lens is able to maintain high quality distance and near vision while enhancing distance vision, which previous multifocals were not able to do. However, multifocal IOLs cannot provide a continuous range of clear vision.
EDOF IOL vs Accommodating IOL
Accommodating IOLs are another type of artificial lens that provides a wide range of clear vision. What sets accommodating lenses apart is their unique mechanism, which mimics the way that eyes naturally focus on objects. This process is called accommodation.
The eye moves and changes shape to allow focus to shift from one distance to another. As the eye ages, it starts losing its ability to “accommodate”. Older ages eventually lose their ability to focus on near objects properly because of this loss of eye flexibility. This condition is called presbyopia, also known as age-related farsightedness.
Besides restoring clear vision, the ultimate goal of premium IOLs like multifocal and EDOF lenses is to correct presbyopia and provide clear near, intermediate and distance vision. Multifocal and EDOF IOLs introduce unique ways to achieve this. But accommodating IOLs provide similar results in a way that feels more natural. However, functionality is limited, as these lenses cannot fully replicate the accommodation process.
Because these lenses rely on certain parts of the eye working as expected (the ciliary muscles and the nodules that are responsible for accommodation), if these parts are compromised, the lenses won’t improve near vision.
EDOF Toric IOL
If a person has astigmatism, cataract surgery or RLE alone won’t correct it. Instead, they will need to use a special IOL called a “toric” lens. When you have astigmatism, the curve of your eye is slightly imperfect. Rays of light entering the eye aren’t focused in one spot on the back of the retina. Instead, they’re spread out and don’t quite hit the retina in the right spot. Toric lenses have a special shape that balances the curve of the eye, correcting astigmatism so that light can focus properly.
Toric technology can be combined with other lens types. This means that people can get toric monofocals, toric multifocals, toric accommodating lenses and even toric EDOF lenses.
Most EDOF IOL models have a toric version. The toric EDOF models provide the same vision outcomes as their non-toric version, with the added benefit of astigmatism correction.
EDOF IOL Cost
The extended depth of focus IOL itself generally falls within the range of $1,500 to $2,500. Popular brands include Alcon’s AcrySof IQ Vivity and TECNIS Symfony, with prices reflecting their advanced optical designs.
Keep in mind that insurance plans won’t cover premium lenses like EDOFs IOLs. If you are interested in enhanced vision outcomes after your cataract surgery or RLE procedure, investing in an EDOF IOL might be worth the cost. At NVISION, we have financing options for patients interested in this lens technology, so you don’t have to pay the entire cost of the lens upfront.
Schedule a consultation at one of our centers to discuss your surgery and IOL options with one of our specialists.
EDOF IOL Brands
There are multiple extended depth of focus IOL models available. Besides having different manufacturers, these lenses utilize different technology to achieve an extended depth of focus. The Acrysof IQ Vivity IOL from Alcon is the more innovative in terms of technology, as it has been designed to overcome the challenges associated with diffractive lenses, like the Tecnis Symfony and Tecnis Synergy, and non-diffractive lenses,like the IC-8 Aphthera.
Your eyecare professional can discuss with you in further detail the pros and cons of EDOF lenses and the various brands available. Keep in mind that surgeon preference and past patients’ satisfaction might impact which EDOF lens or lenses a provider will offer.
You deserve clear vision. We can help.
With 135+ locations and over 2.5 million procedures performed, our board-certified eye surgeons deliver results you can trust.
Your journey to better vision starts here.
References
- FDA Approves First Intraocular Lens with Extended Range of Vision for Cataract Patients. U.S. Food and Drug Administration.
- Matching Color Images: The Effects of Axial Chromatic Aberration. Journal of the Optical Society of America.
- Premium IOLs: How to Spot Poor Candidates. Review of Ophthalmology.
- Indications and Important Safety Information for TECNIS Symfony and TECNIS Symfony Toric Extended Range of Vision IOLs. Johnson & Johnson Vision.
- Refractive Lens Exchange (Lens Replacement Surgery). All About Vision.
- Cataract Surgery. Mayo Clinic.
- Astigmatism. American Optometric Association.
- Facts About Presbyopia. National Eye Institute.
- Presbyopia. All About Vision.
- Facts About Cataracts. National Eye Institute.
- JOHNSON & JOHNSON VISION BRINGS TECNIS SYNERGY AND TECNIS SYNERGY TORIC II PC-IOLS TO NORTH AMERICA FOR CATARACT PATIENTS (June 23, 2021). Johnson & Johnson Vision.
- TECNIS Synergy Intraocular Lens, Tecnis Synergy Toric II IOL, TECNIS Synergy with TECNIS Synergy Delivery System, TECNIS Synergy Toric II IOL with TECNIS Simplicity Delivery System. (July 2021). U.S Food and Drug Administration.
- Alcon Introduces AcrySof IQ PanOptix Trifocal IOL in the U.S., the First and Only FDA-Approved Trifocal Lens (August 2019). Alcon Media Release.
- Alcon launches extended depth of focus intraocular lens. (January 2021). Mass Device.
- Comparison of the Visual Outcomes of an Extended Depth-of-Focus Lens and a Trifocal Lens. (July 2021). Journal of Clinical Ophthalmology.
- The PanOptix Trifocal IOL vs the ReSTOR 2.5 Active Focus and ReSTOR 3.0- Add Multifocal Lenses: A Study of Patient Satisfaction, Visual Disturbances, and Uncorrected Visual Performance. (March 2021). Journal of Clinical Ophthalmology.
- Kohnen T, Berdahl JP, Hong X, Bala C. The Novel Optical Design and Clinical Classification of a Wavefront-Shaping Presbyopia-Correcting Intraocular Lens. Clin Ophthalmol. 2023 Aug 18;17:2449-2457. doi: 10.2147/OPTH.S400083. PMID: 37614847; PMCID: PMC10443698.
- Kanclerz P, Toto F, Grzybowski A, Alio JL. Extended Depth-of-Field Intraocular Lenses: An Update. Asia Pac J Ophthalmol (Phila). 2020 May-Jun;9(3):194-202. doi: 10.1097/APO.0000000000000296. PMID: 32511121; PMCID: PMC7299221.
This content is for informational purposes only. It may have been reviewed by a licensed physician, but is not intended to serve as a substitute for professional medical advice. Always consult your healthcare provider with any health concerns. For more, read our Privacy Policy and Editorial Policy.

